40 days dossier writing for a new benefit assessment
Client: International acting research-based pharmaceutical company
Field of application: Chronic kidney disease
Challenge
Six years after the initial approval, the German organization of a pharmaceutical entrepreneur was surprised by an indication extension for one of its drugs. The AMNOG-experienced team was informed at very short notice, because the international colleagues were not aware that only four weeks after confirmation of the indication extension by the European Commission, a new benefit assessment would be triggered by the Joint Federal Committee (G-BA). This is formally accompanied by the need to submit a new dossier. The dossier had to be written in about 40 days in cooperation with another external cooperation partner in the field of market access.
Solution and approach
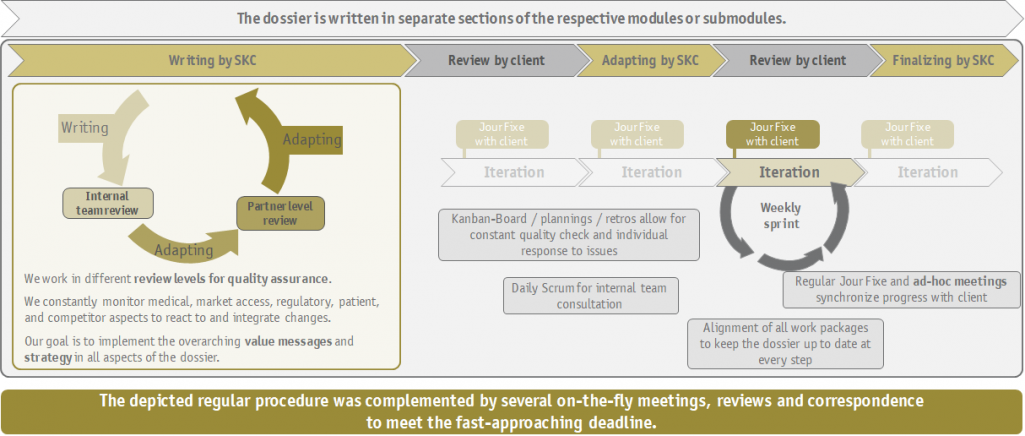
Due to the indication extension, there was already a dossier created by a previous team, on the basis of which we were able to work. This dossier had to be
- revised using a value story that was developed in the agile writing process and
- based on the current state of research;
- updated for the new population;
- built up according to the available evidence for the target population (whereby this evidence, i.e., module 4 and module 5, was compiled by the other cooperation partner).
Meanwhile, a lot of short-term coordination and corresponding adjustments were required, as the usual strategic elaboration prior to the writing process was not feasible in this project. We at SKC benefited from our dossier experiences in a wide variety of indications. In addition, due to the shortage of time and the not fully developed strategy, further "what if...?"-questions were asked of us as strategy experts.
Added value
Despite the high time pressure, the prompt entry into the project and the coordination of three partners involved (consisting of the client, an external dossier team and our team from SKC), excellent cooperation, a lot of flexibility and a convincing effort resulted in a fully comprehensive value dossier that served as the basis for a regular benefit assessment.