Value dossier
The challenge
Establish the foundation
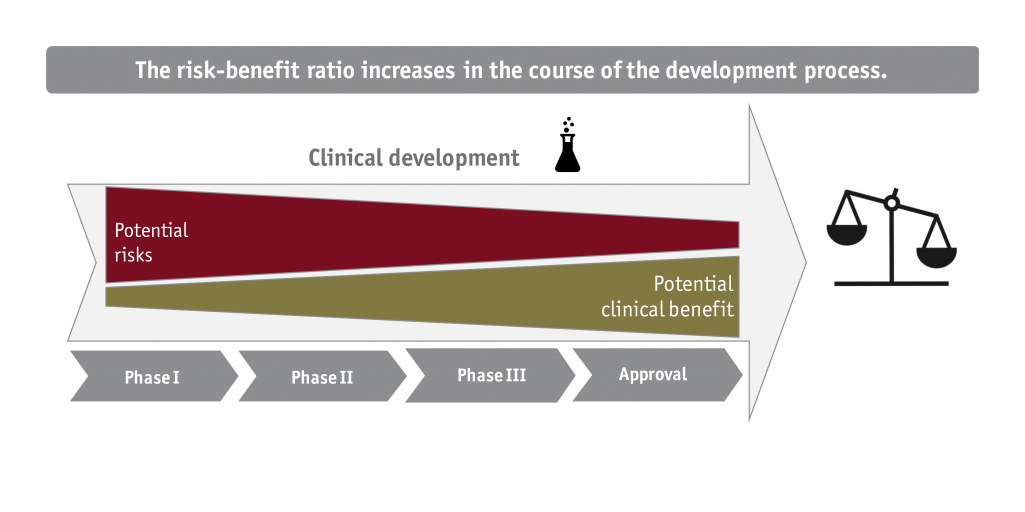
Innovative medication and new medical/biotechnological products face many hurdles before receiving market approval. In order to prove compliance with the legal framework in Germany, the Federal Joint Committee ("Gemeinsamer Bundesausschuss", G-BA) requires the submission of a value dossier. This dossier is the central basis of the value assessment and the subsequent path forward to price negotiations for each product. The core questions for pharmaceutical companies are:
- Which aspects specific for Germany must be considered when creating the value dossier in order to meet all formal requirements?
- What statistical data (endpoints, etc.) are particularly relevant as evidence in order to obtain a positive benefit assessment?
- To what extent can the value story convince all stakeholders participating in the process?
- What does the value dossier need to provide to prove itself during the subsequent AMNOG procedure (statement, hearing, price negotiation, arbitration)?
- How to react flexibly to changing conditions during dossier creation and still ensure a comprehensive and timely submission?
- Which arguments in the value dossier support the highest possible price in Germany as a major European reference country?
The solution
Speak of the added value of the product
The value dossier includes information on areas of application, (comparable) therapies, patient numbers, costs for statutory health insurance providers, study results, etc., and consists of the following five modules:
- Module 1: Administrative information and compilation of statements
- Module 2: General information on the medication and designation of approved areas of application
- Module 3: Designation of the appropriate comparable therapy, number of patients with therapeutically significant benefit, costs of treatment for statutory health insurance providers, requirements for quality assured application
- Module 4: Systematic overview of medical value and medical additional value (description of the methodology and the results), information on patient groups for which there is significant added therapeutic benefit
- Module 5: Literature and documents
Above all else, the purpose of the value dossier is to convincingly argue to the G-BA the benefit of the respective drug or product. Its unique effects must stand in the foreground.
Our approach
Argue convincingly
Together with our clients, we develop a value story that functions as the value driver within your value dossier. It includes, among others, the value story of the product and allows the various stakeholders to be involved in a precise manner. Eventually, a value dossier is created with evidence-based value arguments that are the logical and consistent basis of the entire document and provide a convincing core message. The team at SKC consulting consists of experienced consultants and medical writers so that we can provide, from one source, a value dossier customized to the specific situation.
Agile procedures
The dossier environment is volatile, uncertain, complex, and ambiguous (in short: VUCA). Therefore, the framework of the dossier creation may change constantly due to delays in EMA, fast-track approval, etc., for example. From our many years of experience in consulting, we know how the necessary flexibility, speed, and efficiency can be achieved, especially via an agile organization of labor. For this reason, when creating the dossier we work in agile project teams into which clients are integrated as valuable team members. Short communication paths and a constant flow of information enable us to always be up-to-date and make decisions together. Throughout the process of dossier creation, our clients can rely on our services: whether in the form of a worry-free package or individual milestones during the process.
-
Value Story
Creation of a value story for the strategic direction of the value dossier
-
Strategy
Support and preparation along the value assessment process, e.g. for G-BA consultation (preparation of consultation requirements, communication with the G-BA, participation in counseling sessions) as well as preparation of the registration documents and their supporting evidence
-
Whitepaper workshop
Development of the key points of the module
-
Medical writing
Creation of the entire value dossier or individual modules
-
Review und stress test of the value dossier
Analysis of gaps and implementation of the findings from this, and the analysis of the conditions of other HTA authorities like NICE, HAS
-
Organization
Submission of the dossier to the G-BA and communication with the G-BA
-
Competitor analysis
Preparation and analysis of competitive products as the basis of the product's value
-
Risk Assessment Workshop
Workshop with a systematic risk analysis to identify and assess possible obstacles
Get in touch

Founder and Managing Director
Fax: +49 511 64 68 14 18